
The Global Truth Engine for Better Human Health™
See the world as it truly is — powered by our trusted data, stringent science, and a fusion of AI and human intelligence. TriNetX delivers evidence that stands up to the world’s most rigorous scientific scrutiny.
The world’s most-cited data source in peer-reviewed journals.
Trustworthy Answers in a World Crowded with Doubt
Bring us your toughest medical, scientific, and business questions.
TriNetX connects healthcare organizations and researchers across the globe so they can see what’s happening in real patient populations—not guesses, not simulations.
Our aim is simple and ambitious: Provide a clear path to trustworthy answers, whether your goal is to create promising new therapies, publish groundbreaking research, or improve health outcomes.
We keep data where it belongs — within a secure, connected network of more than 13,000 clinical sites across 20+ countries worldwide — preserving security, provenance, and granularity. Our trusted partnerships ensure high-fidelity data, and a secure, properly governed connection back to our sites and the patients they care for.

Three Strengths, Working Together, Set TriNetX Apart
It’s a combination others can’t match.
Directly-Sourced Data
TriNetX’s data comes from direct partnerships with leading health systems worldwide, not purchased or scraped datasets. Because the data stays within those systems, it remains current, traceable to real patients, and grounded in real care — making it highly reliable and easy to trust. These direct relationships also enable continuous data expansion, custom data curation, and source data validation.
Relentless Integrity
TriNetX stakes its trusted reputation on data quality, security, and scientific integrity. That includes being transparent about our data, our methods, and what use cases the data can — and cannot — support. Our team partners closely with customers to ask the right questions, work through the appropriate scientific methods that match the data, and deliver answers we can all stand behind. We’re also honest enough to say upfront when TriNetX isn’t the right fit.
Fusion of AI and Human Intelligence
TriNetX uses modern, advanced analytics and AI to analyze data and provide actionable insights, not just observations. But people — not technology alone — guide the work. World-renowned scientists and researchers design the studies and scrutinize results to make sure findings make sense in the real world and address the intended use case.
Evidence that Stands Strong Under the World’s Most Rigorous Scientific Scrutiny
TriNetX harmonizes data so it’s easy to use, then supercharges it with machine learning and other powerful tools that surface patterns, accelerate research, and generate real-world evidence in near-real-time. Our world-renowned scientific minds work alongside pharma, healthcare, and academic research customers to frame smarter questions, pinpoint the right data, interpret results, and unearth insights that launch you forward.
Pinpoint the right sites and real patients from the start. See actual patient journeys and patterns of care. And design trials that don’t just withstand scrutiny — they clear the gauntlet.
Bring new drugs and innovative therapies to market faster with trial design built for success with the patient at the center and global, source-verified data that supports regulatory-grade real-world evidence.

Get more from your own data by adding context with purpose-built analytics and global, real-world evidence to inform decisions across research, care delivery, and operations.
Gain deeper insight into patient populations, care pathways, and performance — so you can reduce waste, optimize services, better manage care, and elevate your impact as a trusted research and care partner.

Level-up your research enterprise, fuel competitive grant proposals, and turn classrooms into living labs by giving students hands-on access to global, real-world healthcare data.
With transparent data, scientific support, and global cohorts you can trust, accelerate defensible research from idea to publication with TriNetX — the gold standard for rigorous science.

AI Hype Outstrips its Realities
Others may tell you that AI is a shortcut to perfect answers. But we know AI and other technologies are no better than the quality of the data that feeds them. TriNetX builds AI and machine learning into a disciplined, science-first approach—grounded in strong data, transparent methods, and expert oversight.
Without that scientific foundation, AI can lead you headlong into bad decisions that are costly, time-consuming, and even catastrophic.
What matters isn’t exaggerated promises, but answers and outcomes that withstand scrutiny.
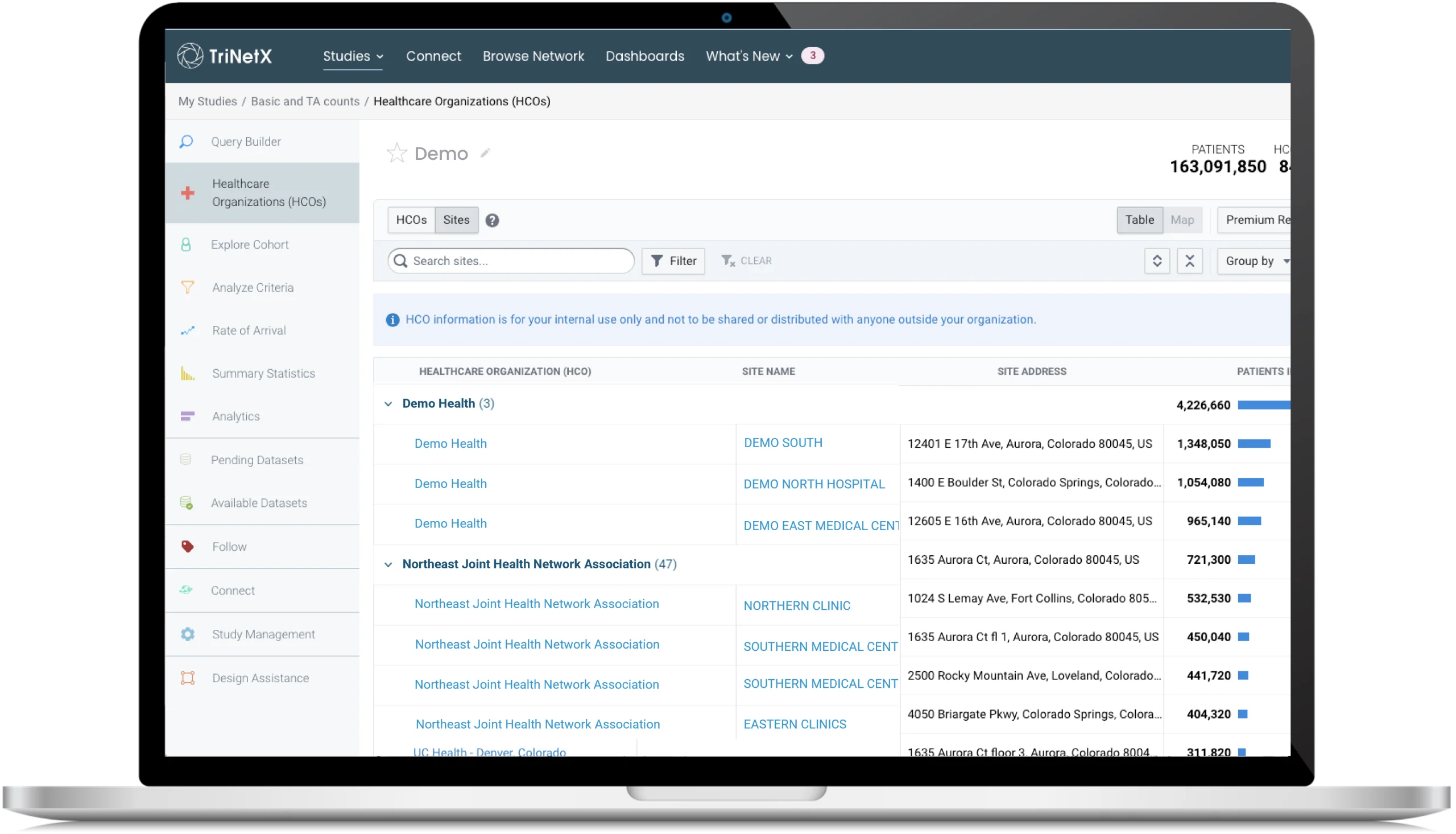
TriNetX Customers Share Real-World Impact
“… what attracted us to the system was the ability to do real-time querying in support of our protocol optimization goals.”
The Global Truth Engine for Better Human Health™
At TriNetX, we enable life sciences, healthcare, and academic institutions to strengthen human health—with data you can trace, evidence you can defend, and answers you can trust.

See What's Shaping Real-World Research Today
The TriNetX LIVE™ network has crossed 300 million patients across 240+ healthcare organizations and 13,000+ sites.

Jeff Brown recently took the podium at ISPOR 2026 to share how we, as a community, can use real-world data to generate robust and actionable evidence.

At ISPOR 2026, we presented posters addressing outcomes in relapsed/refractory DLBCL, and introducing a reproducible method for generating population estimates robust enough to stand up in HTA and regulatory submissions.

We are honored to share that TriNetX has won the “Clinical Trial Innovation Award” in the 10th annual MedTech Breakthrough Awards.
Contact Us
Need to reach out? We’re here to help.
The quickest way to get in touch with the right team is by completing the relevant form below. We will get back to you as soon as possible.
General inquiries
Have a question about our platform, need support, or simply want to learn more about what we do?
Please share your details and we’ll get back to you soon.
"*" indicates required fields
Submit RFI/RFP
Looking to explore a potential partnership or request detailed information about our services?
Send us your request and someone from our team will be in touch.
"*" indicates required fields
Book a demo - TriNetX LIVE™
Book a demo of our real-world data platform for your clinical research.
"*" indicates required fields
Book a demo - EVIDEX®
Book a demo of our powerful, intuitive signal detection and management platform.
"*" indicates required fields
"*" indicates required fields
"*" indicates required fields
"*" indicates required fields
"*" indicates required fields





