Bladder cancer remains a major global health challenge, ranking as the tenth most common cancer overall and the sixth among men. Yet across geographies, the burden of disease looks very different, driven by a complex mix of environmental exposures, diagnostic practices, and access to contemporary treatments.
Within this landscape, perioperative systemic therapy plays a pivotal role in managing muscle-invasive bladder cancer (MIBC), with guideline recommendations placing particular emphasis on neoadjuvant chemotherapy. In this blog, we take a closer look at real-world treatment patterns in systemic perioperative therapy for MIBC and how closely current practice aligns with clinical guidelines.
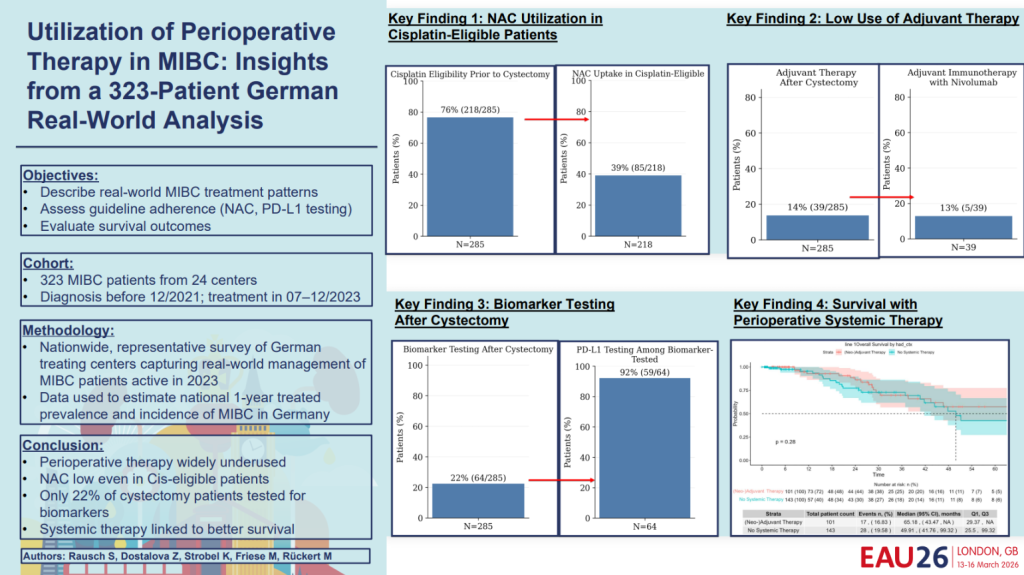
These insights were recently shared at the European Association of Urology conference in London, highlighting real-world evidence from German clinical practice.
Or download a version here >>>
A Closer Look at Real-World Practice in Germany
Our team conducted a retrospective, survey-based, multicenter cohort study including 323 MIBC patients in Germany who were prevalent or incident between July 1 and December 31, 2023. Among them, 73 individuals had progressed from non–muscle-invasive disease.
The study captured a comprehensive view of patient management, including systemic therapy use across the perioperative journey, neoadjuvant, adjuvant, and second-line treatment, as well as PD-L1 testing and survival outcomes. Analyses were stratified by cisplatin eligibility, institution type, and biomarker status.
Twenty-four centers contributed to the survey:
- 3 university hospitals (13%)
- 13 non-university hospitals (54%)
- 8 office-based practices (33%)
Among the 285 MIBC patients undergoing cystectomy, 218 (76%) were eligible for cisplatin. However, only 85 (39%) received neoadjuvant chemotherapy.
Key Real-World Insights
Underutilization of Neoadjuvant Chemotherapy (NAC)
Despite strong support from clinical guidelines, NAC remains markedly underused even among patients who are clearly eligible. This gap between evidence and practice may reflect concerns about toxicity, comorbidities, or logistical challenges that limit the timely initiation of systemic therapy before surgery.
Low Use of Adjuvant Therapy
Adjuvant systemic therapy following radical cystectomy was also infrequent. As a result, many patients, especially those who did not receive NAC, may be missing additional opportunities for disease control in the postoperative period.
Limited Biomarker Testing After Cystectomy
Only around 22% of cystectomy patients underwent biomarker testing such as PD-L1 assessment. As immunotherapy continues to reshape treatment paradigms, limited testing may restrict access to personalized treatment strategies and optimal sequencing of systemic therapies.
Systemic Therapy and Survival Outcomes
Patients who received perioperative systemic therapy demonstrated improved survival outcomes compared with those who did not. These findings reinforce the real-world value of adhering to guideline-directed systemic approaches.
Implications for Clinical Practice
Taken together, these results reveal a substantial implementation gap in the perioperative management of MIBC. While evidence supporting systemic therapy continues to build, adoption in routine practice remains inconsistent. Addressing challenges such as patient selection, multidisciplinary coordination, and timely biomarker testing will be critical to improving outcomes for patients with MIBC.
In summary, perioperative systemic therapy remains significantly underused, even among clinically eligible patients. Enhancing adherence to recommended neoadjuvant treatment and expanding biomarker testing may represent key opportunities to improve survival and elevate the standard of care.
Explore how TriNetX® high-quality real-world evidence can strengthen your research.
About Zuzana Dostalova
Zuzana helps transform real‑world health data into meaningful insights that support better research and patient care. She guides scientific strategy, strengthens data quality standards, and collaborates with global partners to promote responsible, transparent, and impactful use of real‑world evidence.